Diagnosing prostate cancer means finding out whether cancer cells are present in your prostate gland. It starts with screening tests that look for warning signs, then moves to more detailed examinations if needed. The process typically includes a blood test that measures prostate specific antigen (PSA), a physical examination of the prostate, imaging scans like MRI, and ultimately a biopsy where tiny samples of prostate tissue are removed and examined under a microscope. Only a biopsy can confirm whether you have prostate cancer or not.
This article walks you through each step of the diagnostic process, from your first GP appointment to understanding your final results. You’ll learn what each test involves, what the numbers and scores mean, and how doctors decide which tests you need. We explain the difference between screening and diagnosis in the UK, what happens during procedures like the digital rectal exam and prostate biopsy, and how specialists use MRI scans and scoring systems to assess your risk. You’ll also discover who should consider testing, when additional scans might be necessary, and what your results mean for treatment decisions.
Why early diagnosis of prostate cancer matters
Finding prostate cancer early gives you more treatment options and a significantly better chance of successful outcomes. When doctors detect the disease before it spreads beyond the prostate gland, your five-year survival rate approaches 100%. This high survival rate drops substantially once cancer spreads to distant organs or bones, which is why understanding how is prostate cancer diagnosed and acting on symptoms or screening opportunities can literally save your life.
The survival advantage of early detection
Early-stage prostate cancer often grows slowly and remains confined to the prostate gland for years. During this window, you can choose from several effective treatments including surgery, radiotherapy, or even active surveillance if your cancer is low-risk. Treatment at this stage targets a contained area, making it more straightforward to remove or destroy all cancer cells. Once cancer escapes the prostate capsule and spreads to lymph nodes or bones, treatment becomes more complex and focuses on controlling rather than curing the disease.

Catching prostate cancer when it’s still localised means you can access curative treatments with fewer side effects and better long-term health outcomes.
Impact on treatment choices
Your diagnosis timing directly affects which treatments your specialist can offer you. Localised cancers detected early give you the option of robotic prostatectomy, which removes the entire prostate with minimal invasiveness, or targeted radiotherapy that spares surrounding healthy tissue. You might even qualify for active surveillance, where your doctor monitors slow-growing cancer without immediate intervention, sparing you from treatment side effects entirely. Advanced cancers that have spread limit your choices to hormone therapy, chemotherapy, or palliative care. These systemic treatments often come with more significant side effects and cannot eliminate cancer that has already reached distant sites. The difference between early and late diagnosis isn’t just statistical; it’s the difference between potentially curing your cancer and managing it as a chronic condition.
How to approach testing and diagnosis
Your journey to diagnosis begins with an honest conversation with your GP about any symptoms or concerns you have. You don’t need to feel embarrassed about discussing urinary problems, erectile difficulties, or other prostate-related symptoms. Your doctor sees these issues regularly and will take your concerns seriously. If you’re over 50 or have risk factors like a family history of prostate cancer, you can request screening even without symptoms. The key is to take that first step and book an appointment rather than waiting for problems to worsen.
Starting with your GP
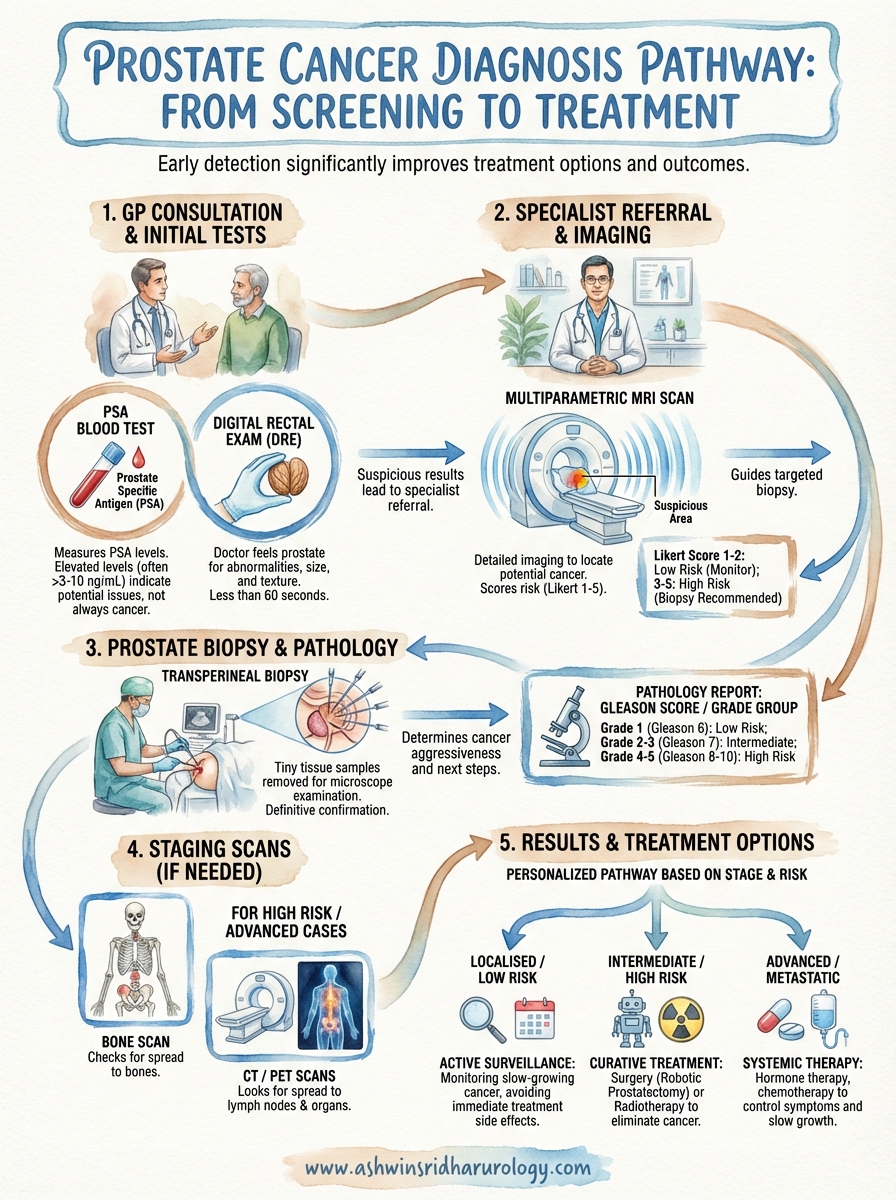
Most men first discuss prostate concerns during a routine GP appointment. Your doctor will ask about your symptoms, how long you’ve had them, and whether anything makes them better or worse. They’ll want to know about your family history, particularly if any close relatives have had prostate cancer. Your GP typically performs a digital rectal examination and orders a PSA blood test as initial screening measures. These simple tests help your doctor decide whether you need referral to a specialist urologist. Don’t skip this step by trying to self-diagnose or requesting specialist referrals directly, as your GP’s assessment provides crucial baseline information that guides the entire diagnostic process.
Understanding the diagnostic pathway
The pathway from suspicion to confirmation follows a clear sequence that protects you from unnecessary procedures. After your initial GP visit, you’ll receive referral to a urologist if your PSA level is elevated or your physical examination raises concerns. Understanding how is prostate cancer diagnosed helps you see that each test serves a specific purpose: PSA testing identifies potential problems, MRI scans locate suspicious areas, and biopsies confirm or rule out cancer. Specialists don’t jump straight to invasive procedures. They build a complete picture using less invasive tests first, only progressing to biopsy when imaging or blood results suggest cancer might be present.
Each diagnostic test adds another piece of evidence, helping your medical team make informed decisions about whether you need further investigation or treatment.
Preparing for your appointments
Preparation makes the diagnostic process smoother and less stressful. Before your appointments, write down your symptoms including when they started, how often they occur, and their severity. List all medications you take, including over-the-counter supplements, as some can affect PSA levels. Bring a family member or friend to important appointments, as they can help you remember information and ask questions you might not think of. Keep a folder with copies of all test results, referral letters, and appointment notes. This organised approach ensures nothing gets overlooked and helps you feel more in control during what can be an anxious time.
Who should consider a prostate cancer check
Understanding who should consider testing helps you make informed decisions about your prostate health. The NHS doesn’t run a national screening programme for prostate cancer because the tests aren’t always accurate and screening can detect slow-growing cancers that might never cause problems. However, you have the right to request a PSA test after discussing the benefits and risks with your GP. Knowing when to seek testing and understanding how is prostate cancer diagnosed empowers you to take control of your health at the right time.
Age-based recommendations
You should consider starting discussions about prostate testing around age 50 if you have no particular risk factors. This age marks the point when your risk of developing prostate cancer begins to increase significantly. Men with a family history of the disease or who are Black should begin these conversations earlier, typically from age 45. Your GP can assess your individual risk and help you decide whether PSA testing makes sense for your situation. Don’t assume you’re too young for prostate problems; some aggressive cancers develop in men in their 40s, particularly those with inherited genetic mutations.
Family history and genetic risk
Family history dramatically increases your need for earlier and more vigilant testing. If your father or brother had prostate cancer, your risk doubles compared to men with no family history. This risk increases further if multiple relatives had the disease or if they were diagnosed before age 60. You should inform your GP about any blood relatives with prostate, breast, or ovarian cancer, as inherited mutations in genes like BRCA1 and BRCA2 affect prostate cancer risk. Genetic counselling might be appropriate if your family has a strong cancer history, helping you understand your specific risk level and surveillance needs.
Black men and ethnicity factors
Black men face twice the risk of developing prostate cancer compared to white men and often develop more aggressive forms of the disease at younger ages. If you’re a Black man, you should discuss testing from age 45 even without symptoms or family history. The reasons for this increased risk aren’t fully understood but likely involve a combination of genetic, environmental, and social factors. Asian men generally have lower prostate cancer rates, but this doesn’t mean you should ignore symptoms or skip discussions about testing if you have other risk factors.
Your ethnic background, family history, and age combine to create your unique risk profile, which should guide when you start considering prostate cancer checks.
Symptoms that warrant immediate checking
Certain symptoms demand prompt medical attention regardless of your age or risk factors. You should contact your GP urgently if you notice blood in your urine or semen, experience bone pain that won’t go away, or find yourself suddenly unable to urinate. Difficulty starting urination, weak urine flow, or needing to rush to the toilet frequently can indicate prostate problems that need investigation. Don’t dismiss these symptoms as normal ageing or wait to see if they improve on their own.
Screening versus diagnosis in the UK
The UK takes a different approach to prostate cancer compared to some other countries. You won’t receive automatic invitations for prostate cancer screening as you do for bowel or breast cancer. The NHS doesn’t run a national screening programme because tests like the PSA can produce false positives and might detect slow-growing cancers that never threaten your life. Instead, the system offers informed choice: you can request a PSA test after discussing the pros and cons with your GP. Understanding how is prostate cancer diagnosed starts with grasping this distinction between screening (looking for potential problems) and diagnosis (confirming whether cancer is actually present).
What screening means in the UK context
Screening refers to testing healthy men without symptoms to look for early signs of prostate cancer. Your GP will explain that PSA tests aren’t perfect; raised levels can result from non-cancerous conditions like prostatitis or benign prostatic enlargement. Equally, some men with normal PSA levels can still have prostate cancer. This uncertainty explains why routine screening isn’t standard. You have the right to request a PSA test from age 50 onwards, or from age 45 if you’re Black or have a family history. Your doctor will ensure you understand what the results might mean and the potential consequences before proceeding with the test.
Screening gives you and your doctor early warning signs, but only further tests can confirm whether cancer is actually present.
The diagnostic journey after screening
Diagnosis begins when your screening tests suggest something might be wrong. Your doctor refers you to a urologist who uses additional tests to build a complete picture. This diagnostic phase includes MRI scans to look for suspicious areas in your prostate, and possibly a biopsy to examine tissue under a microscope. These detailed investigations confirm or rule out cancer definitively. Screening might take five minutes in your GP surgery, whilst diagnosis involves multiple appointments with specialists over several weeks. The process feels more intensive because specialists need concrete evidence before telling you that you have cancer or giving you the reassurance that you don’t.
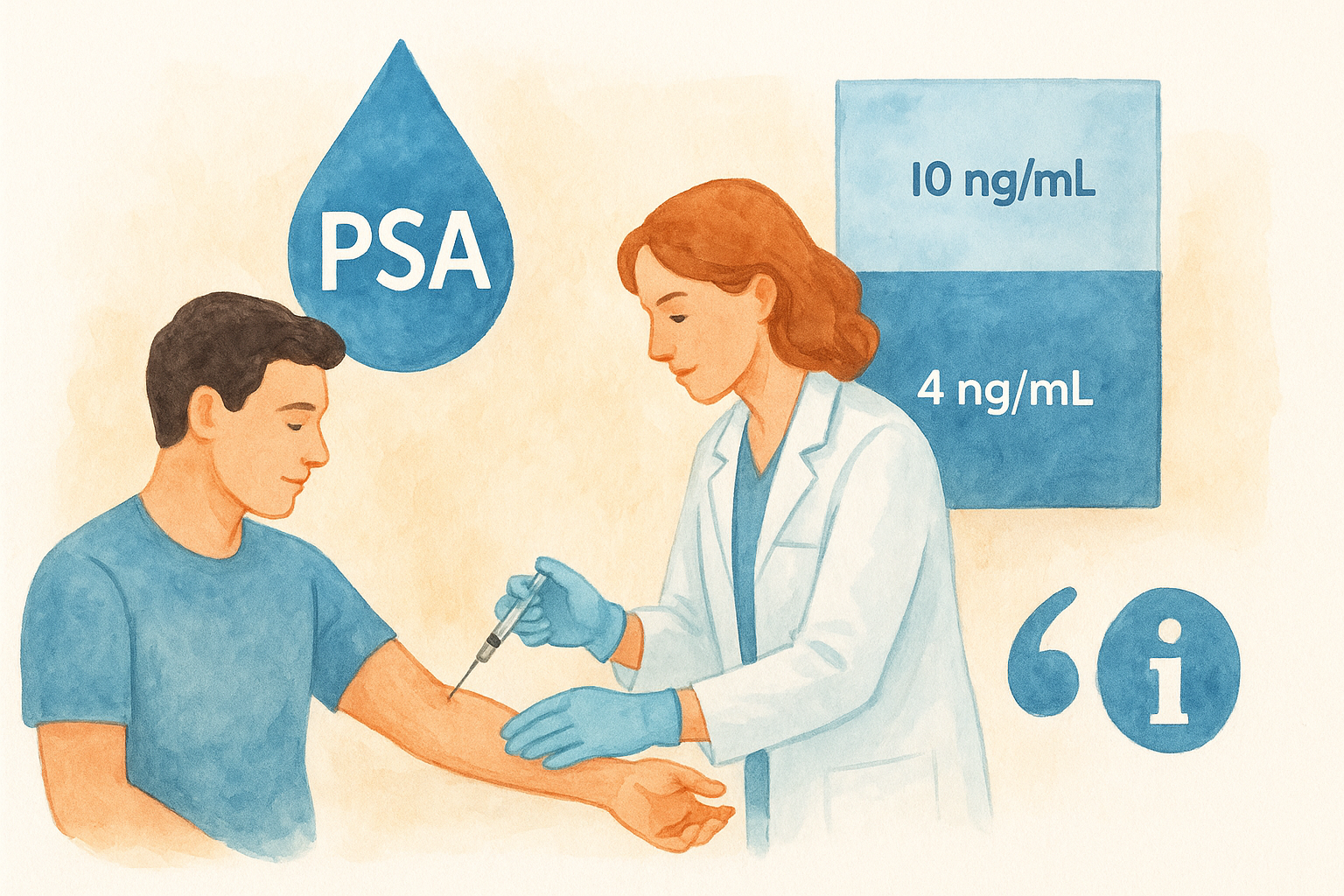
What the PSA blood test tells you
The PSA blood test measures prostate specific antigen, a protein your prostate cells produce. Your prostate releases this protein into your blood in small amounts. When your GP takes a blood sample from your arm, the laboratory measures how many nanograms of PSA appear in each millilitre of your blood (ng/mL). This simple test provides your doctor with valuable information about your prostate health, though it cannot diagnose cancer on its own. Understanding what PSA levels mean helps you grasp how is prostate cancer diagnosed and why doctors might recommend further tests.
What PSA measures and why it matters
PSA exists in your bloodstream naturally, with most men having levels below 4 ng/mL. Your prostate releases more PSA into your blood when something irritates or damages it. Cancer cells often produce higher amounts of PSA than healthy cells, which is why doctors use this test as a warning sign. However, benign conditions like prostate enlargement or infection also raise PSA levels, making the test a useful indicator but not a definitive diagnosis tool. Your doctor looks at your PSA number alongside other factors like your age, prostate size, and symptoms to assess your overall risk.
Higher PSA levels increase your cancer risk, but the relationship isn’t straightforward. Men with PSA between 4 and 10 ng/mL have roughly a one in four chance of harbouring cancer, though many of these prove to be slow-growing tumours that might never cause problems. When your PSA exceeds 10 ng/mL, your cancer risk rises above 50%, prompting most doctors to recommend further investigation with imaging and possibly biopsy.
Your PSA number provides context rather than a definite answer, which is why your doctor always considers it alongside other test results and your personal risk factors.
Factors that affect your PSA level
Several non-cancerous conditions can push your PSA higher, leading to false alarms. Benign prostatic enlargement, a normal part of ageing for many men, causes your prostate to produce more PSA simply because you have more prostate tissue. Prostatitis (prostate inflammation) or urinary tract infections irritate your prostate, temporarily spiking PSA levels. You should avoid ejaculation for 48 hours before your test, as sexual activity can artificially raise PSA. Vigorous exercise, particularly cycling, can also affect results.
Your doctor considers your age and prostate size when interpreting PSA results. Older men naturally have higher baseline PSA levels because prostates typically enlarge with age. Some specialists calculate PSA density by dividing your PSA level by your prostate volume measured on ultrasound or MRI. This calculation helps distinguish between PSA elevation from benign enlargement versus potentially cancerous growth.
What your PSA number means for next steps
Your GP uses PSA results to decide whether you need referral to a specialist. Normal PSA levels below 3 ng/mL typically mean you can continue routine monitoring through your GP, though this depends on your age and other risk factors. Borderline results between 3 and 10 ng/mL might prompt your doctor to repeat the test in a few weeks, checking whether the level stays stable or continues rising. Consistently elevated PSA usually leads to urology referral for MRI imaging and possible biopsy. Rising PSA over time, even if still technically in the normal range, concerns doctors more than a single elevated reading, as upward trends suggest something actively changing in your prostate.
What happens during a digital rectal exam
The digital rectal examination, commonly called a DRE, involves your doctor gently inserting a gloved, lubricated finger into your rectum to feel your prostate gland. Your prostate sits directly in front of your rectal wall, making it accessible through this brief physical examination. The test takes less than a minute and forms a crucial part of understanding how is prostate cancer diagnosed, as your doctor can detect abnormalities that blood tests might miss.
The examination procedure
Your doctor asks you to either lie on your side with your knees drawn up towards your chest, or to stand and bend forward over the examination table. They put on a glove, apply lubricant to their finger, and gently insert it into your rectum. You’ll feel pressure and fullness but shouldn’t experience sharp pain. Your doctor moves their finger carefully to feel different parts of your prostate through the thin rectal wall. The entire examination lasts 30 to 60 seconds, and you can resume normal activities immediately afterwards.
The brief discomfort of a DRE is vastly outweighed by its ability to detect potentially serious prostate changes early.
What your doctor feels for
During the examination, your doctor assesses your prostate’s size, shape, and texture. A healthy prostate feels smooth and relatively soft, roughly the size of a walnut. They check for any hard lumps, bumps, or irregular areas that might indicate cancer. Your doctor also notes whether abnormalities appear on one side of the prostate or both, and whether the gland feels tender, which might suggest infection rather than cancer. Asymmetry, hardness, or nodules prompt further investigation with imaging scans and possible biopsy. Your doctor combines DRE findings with your PSA results to build a complete picture of your prostate health.
MRI of the prostate and Likert scoring
Magnetic resonance imaging (MRI) scans create detailed pictures of your prostate using magnetic fields and radio waves rather than radiation. Your specialist typically orders an MRI after your PSA test or DRE suggests potential problems, but before proceeding to a biopsy. This imaging test helps doctors locate suspicious areas within your prostate that might contain cancer, making any subsequent biopsy far more accurate. The multiparametric MRI (mpMRI) used for prostate assessment takes multiple types of images to build a comprehensive view of your prostate’s structure and any abnormalities within it.
What MRI shows and why it matters
Your MRI scan helps your urologist see inside your prostate without needing surgery or needles. The scan identifies regions that look different from normal prostate tissue, highlighting areas where cancer might be growing. Radiologists examine the images looking for changes in tissue density, blood flow patterns, and cellular behaviour that suggest malignancy. This technology proves particularly valuable because it can detect clinically significant cancers whilst helping doctors avoid finding tiny, slow-growing tumours that might never threaten your health.
MRI plays a crucial role in modern prostate cancer pathways by reducing unnecessary biopsies. Before widespread MRI use, men with elevated PSA levels often went straight to biopsy, which meant taking multiple random samples from the prostate hoping to hit any cancer present. Now, your specialist uses MRI to see whether suspicious areas exist and precisely where they’re located, meaning biopsies can target specific regions rather than sampling blindly. If your MRI appears completely normal, your doctor might decide you don’t need a biopsy at all.
MRI transforms how is prostate cancer diagnosed by allowing doctors to see potential tumours before taking tissue samples, making the entire process more accurate and less invasive.
Understanding Likert scores and PI-RADS
Radiologists grade your MRI findings using the Likert scale or its standardised version called PI-RADS (Prostate Imaging Reporting and Data System). These scoring systems range from 1 to 5, estimating how likely any abnormal areas are to contain clinically significant cancer. A score of 1 or 2 suggests cancer is highly unlikely, whilst scores of 4 or 5 indicate cancer is probably present. The score of 3 sits in the middle, representing uncertainty where cancer might or might not exist. Your radiologist examines multiple MRI sequences showing different tissue properties, then combines these observations into a single score that summarises your risk level.
PI-RADS provides a standardised language that all specialists understand, ensuring consistent interpretation regardless of which hospital or radiologist reviews your scan. The system helps your urologist make evidence-based decisions about whether you need a biopsy and which areas of your prostate warrant the closest attention during that procedure.
What your MRI score means for next steps
Your MRI results determine your next steps in the diagnostic pathway. Scores of 1 or 2 typically mean you avoid biopsy, though your doctor considers other factors like PSA density and your overall risk profile before making final recommendations. You’ll usually return to regular monitoring through your GP with repeat PSA tests every six to twelve months. Some men with borderline results might have a repeat MRI after a period of watchful waiting to see whether any changes develop.
Scores of 3, 4, or 5 usually lead to a targeted biopsy where your urologist samples the suspicious areas identified on your MRI. Higher scores prompt more thorough sampling because the likelihood of finding significant cancer increases. Your specialist combines your MRI findings with your PSA level, age, and general health to create a complete picture that guides whether biopsy proceeds immediately or whether additional monitoring might be appropriate in your specific circumstances.
Prostate biopsy, Gleason score and Grade Group
A prostate biopsy remains the only definitive way to confirm whether you have prostate cancer. This procedure involves removing small tissue samples from your prostate using a thin needle, which your specialist then sends to a laboratory for microscopic examination. Your urologist recommends biopsy when your PSA test, DRE, or MRI scan suggests cancer might be present, and understanding how is prostate cancer diagnosed means recognising that biopsy provides the final confirmation that other tests cannot deliver. The procedure typically happens as a day case in hospital or clinic, taking around 15 to 20 minutes from start to finish.
The biopsy procedure and what to expect
Most UK hospitals now use the transperineal approach, where your urologist inserts the biopsy needle through the skin between your scrotum and anus (the perineum) rather than through your rectum. You’ll receive either local anaesthetic to numb the area or a general anaesthetic where you sleep through the procedure. Your specialist uses ultrasound or MRI images to guide the needle to specific areas identified on your earlier scans, taking between 12 and 20 tiny core samples from different parts of your prostate. Targeted biopsies focus on suspicious regions shown on MRI, whilst systematic samples come from across the entire gland to ensure nothing gets missed.
Recovery usually takes a few days, during which you might notice blood in your urine or semen, and your perineum may feel tender. Most men return to normal activities within 48 hours, though you should avoid strenuous exercise for about a week. Your GP prescribes antibiotics to prevent infection, which you start taking before the procedure. Serious complications remain rare, affecting fewer than 2% of men who have the procedure.
Understanding your Gleason score
Pathologists examine your biopsy samples under microscopes to look for cancer cells and, if present, determine how aggressive they appear. The Gleason scoring system grades cancer cells on a scale from 1 to 5 based on how much they differ from normal prostate tissue. Grade 1 cells look almost normal and grow slowly, whilst grade 5 cells look very abnormal and tend to grow and spread quickly. Pathologists identify the two most common patterns in your samples, then add these numbers together to create your Gleason score.

Your Gleason score ranges from 6 to 10 in practice, though theoretically it could go as low as 2. A score of 6 (3+3) represents the lowest-grade cancer typically reported and suggests slow growth. Scores of 7 split into two groups: 3+4=7 (where grade 3 predominates) carries better outlook than 4+3=7 (where more aggressive grade 4 cells dominate). Scores of 8, 9, or 10 indicate high-grade cancers that require prompt treatment.
Your Gleason score directly influences your treatment options, with higher scores typically requiring more aggressive intervention to control faster-growing cancers.
Grade Groups and treatment planning
Modern pathology reports increasingly use Grade Groups alongside or instead of Gleason scores because they communicate risk more clearly. This newer system divides prostate cancers into five groups, running from Grade Group 1 (lowest risk) to Grade Group 5 (highest risk). Grade Group 1 corresponds to Gleason 6, Grade Groups 2 and 3 represent the two types of Gleason 7 (3+4 and 4+3 respectively), Grade Group 4 equals Gleason 8, and Grade Group 5 covers Gleason 9 and 10 cancers.
Your Grade Group helps your specialist recommend appropriate treatment by indicating how likely your cancer is to grow and spread. Grade Group 1 cancers might suit active surveillance where doctors monitor rather than immediately treat your cancer, sparing you from treatment side effects. Higher Grade Groups typically require definitive treatment through surgery or radiotherapy because these cancers pose greater risk of progression if left untreated. Your urologist considers your Grade Group alongside your PSA level, MRI findings, and overall health when creating your personalised treatment plan.
Scans and tests that look for spread
Your specialist orders additional imaging tests when your biopsy confirms cancer and they need to determine whether it has spread beyond your prostate. These staging scans look for cancer in your lymph nodes, bones, and other organs. Not everyone diagnosed with prostate cancer needs these tests; your urologist considers your PSA level, Grade Group, and examination findings to decide whether staging scans are necessary. Men with low-risk, early-stage cancers confined to the prostate typically skip staging scans because their cancer is extremely unlikely to have spread. Understanding how is prostate cancer diagnosed includes recognising that staging forms a separate step after diagnosis, painting the complete picture of your disease extent.
When additional imaging becomes necessary
Your doctor recommends staging scans when your cancer shows higher-risk features that suggest possible spread. High PSA levels above 20 ng/mL, Grade Groups of 4 or 5, or abnormal findings on your DRE all increase the likelihood that cancer has escaped your prostate. Specialists also order scans if your symptoms suggest metastatic disease, such as persistent bone pain or unexplained weight loss. Lower-risk cancers with PSA under 10 ng/mL, Grade Group 1 or 2, and no concerning symptoms rarely require staging investigations because they almost always remain confined to the prostate gland.
Bone scans and skeletal imaging
Bone scans help detect whether prostate cancer has spread to your skeleton, which represents the most common site of distant spread. You receive an injection of a low-level radioactive tracer that travels through your bloodstream and accumulates in areas where bone is actively changing or damaged. A special camera then creates images of your entire skeleton two to four hours later. Areas where cancer has spread appear as darker spots on the scan, though arthritis and old injuries can create similar appearances. Your doctor might request additional X-rays or MRI scans to confirm whether suspicious areas truly represent cancer spread or benign bone changes.
Staging scans reveal whether your cancer remains localised or has spread, fundamentally shaping which treatments can cure your disease versus which aim to control it.
CT and PET scans for distant spread
CT (computed tomography) scans use X-rays and computer technology to create detailed cross-sectional images of your body, particularly useful for examining lymph nodes in your pelvis and abdomen. These scans show whether nodes appear enlarged or abnormal, suggesting possible cancer spread. PET (positron emission tomography) scans inject a radioactive tracer that concentrates in cancer cells, creating images that highlight metabolically active areas throughout your body. PSMA PET scans use tracers that specifically target prostate-specific membrane antigen, a protein abundant on prostate cancer cells, making them highly accurate for detecting prostate cancer spread. Your specialist might recommend PSMA PET imaging if your cancer has already spread or if conventional scans give unclear results.
Understanding your results and treatment options
Your test results arrive as a detailed pathology report that combines all the information gathered during your diagnostic journey. This document includes your PSA level, Gleason score or Grade Group, MRI findings, and biopsy results, along with details about how much of your prostate contains cancer and whether it has spread beyond the gland. Your urologist uses these results to assign your cancer a stage and risk category, which directly determines which treatments suit your situation. Understanding how is prostate cancer diagnosed helps you grasp why each piece of information in your report matters when planning treatment.
Reading your pathology report
Your pathology report tells you several crucial facts about your cancer. The percentage of positive biopsy cores (for example, "6 out of 12 cores contain cancer") indicates how widespread the cancer is within your prostate. The report notes whether cancer appears on one side of your prostate or both, and whether it has grown beyond the prostate capsule into surrounding tissue. You’ll see your Grade Group clearly stated, along with the corresponding Gleason score. The report might mention perineural invasion, which means cancer cells have grown along nerve pathways, or extraprostatic extension, indicating cancer has breached the prostate’s outer layer. These technical terms sound alarming, but your specialist explains exactly what they mean for your specific prognosis and treatment needs.
How staging determines your treatment path
Staging combines all your test results into a single classification that describes your cancer’s extent. Stage T1 or T2 cancers remain confined to your prostate, whilst T3 cancers have grown through the prostate capsule, and T4 cancers invade nearby structures like your bladder or rectum. The presence of cancer in lymph nodes moves you to stage N1, whilst distant spread to bones or organs represents M1 or stage 4 disease. Your specialist also considers your PSA level and Grade Group when determining your overall stage, creating categories that range from very low-risk localised cancer to high-risk or advanced disease.
Your cancer stage determines whether treatment aims to cure your disease or control its symptoms, fundamentally shaping your treatment journey and expected outcomes.
Treatment options vary dramatically based on stage. Localised cancers confined to your prostate give you curative options including robotic prostatectomy, external beam radiotherapy, or brachytherapy (internal radiation). You might qualify for active surveillance if your cancer grows slowly and carries low risk of progression. Advanced cancers that have spread require systemic treatments like hormone therapy, which slows cancer growth throughout your body, often combined with chemotherapy or newer targeted treatments. Your urologist discusses each option’s benefits, risks, and potential side effects, helping you choose the approach that aligns with your priorities.
Treatment choices for different risk groups
Your risk group classification simplifies complex staging information into practical categories. Low-risk cancers (Grade Group 1, PSA under 10 ng/mL, stage T1 or T2a) often suit active surveillance or single-modality treatment like surgery or radiotherapy alone. Intermediate-risk cancers might require combination approaches, such as radiotherapy plus a short course of hormone therapy. High-risk localised cancers typically need aggressive multimodal treatment combining surgery or radiotherapy with extended hormone therapy. Your age, overall health, and personal preferences influence which option your specialist recommends, as treatments carry different side effect profiles affecting urinary control, sexual function, and bowel habits. Your specialist ensures you understand the trade-offs between treatment intensity and preserving quality of life.
Next steps and support
You now understand how is prostate cancer diagnosed, from initial PSA testing through to biopsy results and staging scans. This knowledge empowers you to have informed conversations with your GP about when testing makes sense for your situation, and what each result means for your health. The diagnostic process can feel overwhelming, but breaking it into clear steps helps you approach each stage with confidence rather than anxiety.
Your next action depends on where you are in your journey. Men without symptoms or elevated risk might simply discuss screening options with their GP at their next routine appointment. Those with concerning symptoms should book a GP appointment promptly rather than waiting. If you’ve received abnormal test results or a cancer diagnosis, seeking specialist advice becomes the priority. Book a consultation with an experienced urologist who can review your results, explain your options, and guide you towards the treatment approach that best suits your circumstances and priorities.
